Iron that is either near salt water, or areas where roads are salted, rusts relatively quickly compared to metals in other environments. Part of the correct answer is that increasing salt concentration increases the rate of rusting. To answer this question, we need to determine whether it increases or decreases the rate of reaction and the mechanism behind that change. This question is asking how and why salt changes the rate of reaction of rusting. So, the correct answer to this part of the question is “electrons.” During oxidation, iron gives up electrons to form iron 2+ ions. Oxidation involves a loss of electrons, while reduction involves a gain of electrons. Oxidation–reduction reactions involve the transfer of electrons from one compound or element to another. This question is asking about the process of oxidation. Which term best describes the role of the salt solution in the rusting process?.The rate increases because dissolved ions react with the metal atoms.The rate decreases because dissolved ions react with dissolved oxygen.The rate decreases because dissolved ions aid the ionization of water.The rate increases because dissolved ions aid the movement of electrons.The rate increases because dissolved ions aid the decay of metal nuclei.How and why does the rate of rusting of iron in water vary with increasing salt concentration?.Which particles are removed from a metal during an oxidation reaction?.The rate of rusting of iron in water varies with increasing salt concentration. Rusting of iron is an example of a redox reaction. The ions present in salt water make it a more effective electrolyte than fresh water, allowing electrons to be transferred more easily and rust to form more quickly.Įxample 2: Describing the Effect of Salt on Rusting Processes The oxidation–reduction reaction at the beginning of the rusting process requires the movement of electrons. Interestingly, exposure to salt water increases the rate of rusting compared to fresh water. The iron in objects near the sea, such as boats and chains, also tends to rust quite quickly, as can be seen in the photo below. Conversely, if we leave an iron object outside in the rain for many days, it will rust more quickly than if it is kept dry. For example, if we coat the iron in grease so water and oxygen cannot reach it, no rust will develop. The simplest way to affect the rate of reaction is to change the exposure to the two main reactants, water and oxygen. With this process in mind, we can take a look at some of the factors that might increase the rate of rusting of a piece of iron. The iron(III) ions then combine with water to make iron(III) hydroxide, which then forms hydrated iron(III) oxide.
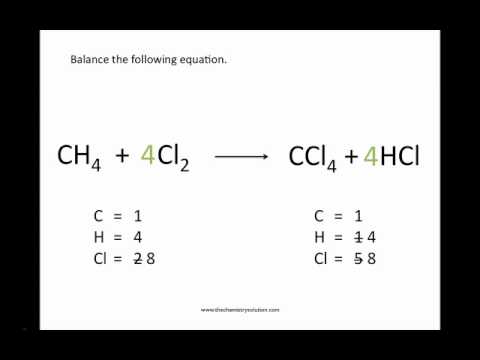
Hydrogen ions are absorbed, and water is produced along the way.
In summary, iron dissolves in water to form iron(II) ions that are then oxidized into iron(III) ions. The iron(III) ions combine with water to form iron(III) hydroxide:į e ( ) + 3 H O ( ) F e ( O H ) ( ) + 3 H ( ) 3 + 2 3 + a q l s a qįinally, the iron(III) hydroxide dehydrates to form hydrated iron(III) oxide with chemical formula F e O H O 2 3 2 ⋅ ?. In the corresponding half reaction, oxygen is reduced, accepting electrons from the reaction above in the presence of hydrogen ions to form water:Ĥ e + 4 H ( ) + O ( ) 2 H O ( ) – + 2 2 a q g lĪs well as reacting to form water, the hydrogen ions and the dissolved oxygen in the water further oxidize iron(II) ions into iron(III) ions:Ĥ F e ( ) + 4 H ( ) + O ( ) 4 F e ( ) + 2 H O ( ) 2 + + 2 3 + 2 a q a q a q a q l Oxidation is the loss of electrons, and this formation of ions happens as the solid iron becomes a solution: The first step is the oxidation of iron to iron(II) ions, as shown by the following half reaction.

However, to understand the chemical process in more detail, let’s look at the intermediate reactions. This overall reaction shows that iron combines with oxygen and water to form a hydrated oxide. The simplified reaction for the formation of rust is:Ĥ F e + 3 O + 2 H O 2 F e O 2 H O 2 2 2 3 2 ? ⋅ ? The “ ?” signifies that the number of water molecules in the compound can vary. Other metals can oxidize or otherwise corrode to form various compounds, but only iron will form the compound we call “rust.”Ĭhemically, rust is hydrated iron(III) oxide, with the chemical formula F e O H O 2 3 2 ⋅ ?. Note that all rust is corrosion, but not all corrosion is rust. Corrosion is the gradual destruction or damage caused by a slow, irreversible, and spontaneous redox reaction between the surface of a substance and the environment.
